Latest articles
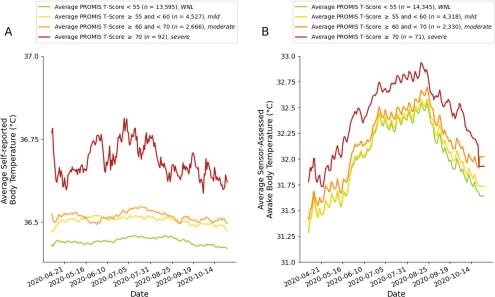
Researchers at UCSF and UCSD show link between body temperature and depression
With support from USAMRDC’s Military Infectious Diseases Research Program (MIDRP), [...]
Developing a Burn Patient Transfer System to Coordinate Emergency Response between Civilian and Government Hospitals
With support from Combat Casualty Care Research Program and MTEC, [...]
FDA Test
Company : BD (Becton Dickinson and Company) Military Sponsor: DDDI/COVID-19 [...]
Developing EyeAid™ to salvage of eye injuries on the battlefield
With support from the US Army Medical Material Development Activity [...]